It is necessary for any concrete surface to be covered with suitable coating to remain protected from attack of conditions which can deteriorate that concrete member. The situation is worse in chemical manufacturing plants where the majority of chemicals are not resisted by concrete due to many reasons.
Corrosive chemicals are usually categorized in “mild’, ‘moderate’ & ‘severe’ categories according to their corrosive index. Mild chemicals coming in contact with concrete require normal protective coating to prevent it from getting deteriorated with exposure of concrete members to those chemicals and similarly severe categories of chemicals require stubborn and sustainable protective coating to ensure their long life even when they are exposed to severely corrosive chemicals.
Caustic chlorine manufacturing plants generate effluent which normally contains a combination of many corrosive chemicals because all of them take part in the manufacturing process of the end product for those Organizations.
Caustic is NaOH which is alkaline in nature and hence its chemical nature is similar to that of concrete. Chlorine evolves as a byproduct during the manufacturing process of the chemical produced at the end. This phenomenon also involves generation of bromine on route. Both chlorine / chloride and bromine / bromide are extremely corrosive for concrete and both of them are present on the effluent generated in the manufacturing unit.
It has been observed in many units manufacturing caustic / chlorine based chemical derivatives that effluent pits are provided with coal tar epoxy based coating. It is also observed that performance of this coating has few serious limitations and hence they are not successful for protecting concrete for a long time.
Coal Tar epoxy is a derivative from basic epoxy material. Chemical formula of bisphenol (epoxy) is C15H16O2 and is having matrix as placed below
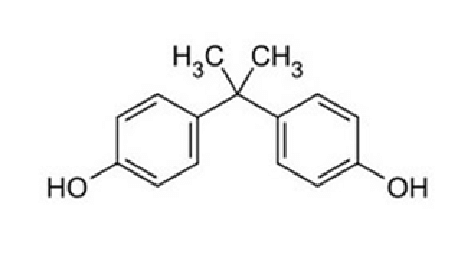
Bisphenol is 4,4′ – methyl-diyldi-phenol in which the methylene hydrogens are replaced by two methyl groups. It has a role as a xenoestrogen, an environmental contaminant, a xenobiotic and an endocrine disruptor. This molecular matrix is again blended with coaltar which contains naphthalene and aromatic hydrocarbons. That being a physical blending and not a chemically reacted and saturated product, it carries free carbon, and aromatic hydrocarbons in the coating.
When effluent containing caustic and chlorine comes in contact with a coating having free carbon and aromatic hydrocarbons, they have a tendency to react because of the high molecular weight of chlorine / bromine and caustic as compared to free carbon and aromatic hydrocarbons. End product at the end of such reactions largely depends on surrounding temperature / humidity. End product which gets generated when effluent comes in contact with coal tar epoxy coating is either carbolic acid or hydrochloric acid gets or sodium carbide. Sodium carbide is an unsaturated matrix which is highly active. Formation of acidic chemical in the middle of alkaline coating starts attacking on the hydrocarbon chain of bisphenol as shown above. Weakening of this chain is then damaged by sodium carbide. This results in failure of continuity of protective chain of coal tar based bisphenol and the performance of coal tar epoxy based protective coatings for effluent tanks of concrete material doesn’t give success here.
Authored By:








